Adaptive Resilience Reserves (ARR) for Pharmaceutical Continuity

Introduction
The Adaptive Resilience Reserve (ARR) framework has been introduced as a structured approach to building continuity under volatility. While earlier applications focused on financial and operational resilience, uninterrupted access to medicines represents one of the most critical resilience challenges for healthcare systems.
Recent global disruptions, particularly the COVID-19 pandemic, demonstrated that medicine availability cannot be assumed. Supply concentration, export restrictions, logistics bottlenecks, and sudden demand surges exposed structural weaknesses in pharmaceutical supply chains. These lessons underline the need to move from reactive stockholding to an adaptive reserve model.
Regulatory Context
Federal Decree-Law No. 38 of 2024 on the regulation of pharmaceutical products establishes a clear policy direction: executive regulations shall define conditions to ensure the provision of necessary pharmaceutical products and medical supplies to meet community needs on a permanent basis. This elevates medicine availability from an operational objective to a governance and compliance requirement. ARR provides a practical mechanism to operate this obligation.
The Current Operating Reality: Why Medicine Resilience Is Now a Strategic Priority
Pharmaceutical supply chains operate under structural constraints that make them uniquely vulnerable to disruption. Unlike other commodities, medicines are subject to strict regulatory controls, cold-chain requirements, limited manufacturing sources, finite shelf lives, and long lead times for replenishment.
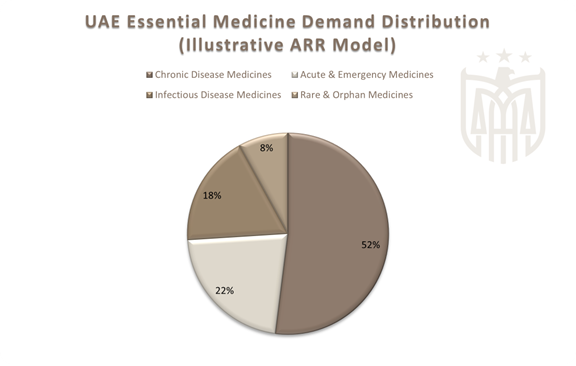
In the UAE, medicine demand is structurally driven rather than cyclical, with chronic conditions such as diabetes, cardiovascular disease, and respiratory illnesses requiring uninterrupted long-term treatment. Rare and orphan medicines further increase supply sensitivity due to limited global production and complex procurement requirements. In this context, traditional minimum stock practices and short-term forecasting are no longer sufficient, reinforcing the need for a structured and adaptive approach to medicine reserves.
Why Pharmaceutical ARR Is Necessary?
Pharmaceutical supply chains face structural constraints including long production lead times, cold-chain requirements, limited manufacturing sources, and finite shelf lives. In the UAE, demand for medicines particularly for chronic conditions such as diabetes is continuous and non-deferrable. Traditional minimum-stock models are insufficient. An adaptive, indicator-driven reserve is required.
Learning from COVID-19:
The COVID-19 pandemic demonstrated several hard truths:
- Global pharmaceutical supply chains are highly concentrated and interdependent
- Export controls and national prioritisation measures can override commercial contracts
- Sudden demand spikes can overwhelm production and distribution capacity
- Reactive stockpiling leads to inefficiencies, expiries, and inequitable access
Most importantly, it showed that emergency responses, while necessary, are inherently inefficient. Resilience cannot be built during a crisis; it must be engineered in advance.
ARR offers a mechanism to institutionalise these lessons, shifting medicine availability from an emergency response function to a permanent resilience capability.
The Adaptive Medicine Reserve Model
Under ARR, medicines are managed as rotating resilience reserves, dynamically calibrated using predefined indicators such as consumption patterns, supplier lead times, logistics disruptions, and public health alerts. When stress indicators rise, reserve coverage expands. When stability returns, stock is rotated through normal channels to reduce expiry risk.
Tiered Structure of Medicine Reserves
Tier 1 – Essential High-Volume Medicines
Medicines required daily by large segments of the population for chronic disease management.
Objective: continuous availability with elevated baseline reserves.
Tier 2 – Critical and Acute Medicines
Medicines where shortages have immediate clinical consequences, including emergency and ICU treatments.
Objective: higher reserve thresholds and rapid activation.
Tier 3 – Rare and Orphan Medicines
Low-volume, high-impact medicines with limited global supply.
Objective: minimum viable stock combined with guaranteed replenishment pathways.
Adaptive Triggers: How the Reserve Responds to Stress
ARR operates through predefined indicators that continuously assess system pressure, including:
- Changes in dispensing and consumption rates
- Expansion of supplier lead times
- Import clearance delays
- Recall frequency and quality alerts
- Cold-chain disruption risks
- Epidemiological or public health alerts
When thresholds are breached, reserve coverage automatically increases. When stability returns, excess stock is rotated back into standard distribution, minimising waste and expiry risk.
This adaptive logic transforms medicine reserves from dormant stock into a living system aligned with real-world conditions.
Governance and Institutional Alignment
For pharmaceutical ARR to function effectively, governance must be clearly defined.
- Manufacturers support continuity through production planning and availability commitments
- Importers and distributors manage reserve stock, rotation, and logistics
- Healthcare providers contribute demand intelligence and clinical forecasting
- Regulators enable oversight, policy alignment, and system-wide visibility
Crucially, ARR does not replace existing regulatory frameworks; it operationalises them. It provides the mechanism through which policy objectives such as continuous availability, patient protection, and system stability are practically achieved.
Conclusion
Pharmaceutical continuity is a matter of national resilience, regulatory compliance, and public trust. By applying ARR to essential and critical medicines, healthcare systems can ensure continuous access, reduce emergency dependency, and align operations with legislative intent.
Adaptive medicine reserves transform stock from a passive buffer into an active resilience instrument supporting long-term healthcare stability and national preparedness.
Copyright: Doctor Mohammed Hassan Al Raeesi Advocates & Legal Consultants retains all intellectual property rights to this content. No third party may use, copy, or modify any part of it without prior permission and without proper attribution to our firm.
Disclaimer: This is a conceptual framework intended for thought leadership and does not constitute legal or financial advice. For professional evaluation of your company’s legal, financial and ESG governance policies, please contact our firm.
📍 1105, Tameem House, Al Barsha Heights, Dubai, UAE
📞 +971 4 298 0686
💬 WhatsApp: +971 56 111 3928
📩 info@dralraeesilegal.com
🌐 www.dralraeesilegal.com
